
Colonoscopy, which is the gold standard for the assessment of inflammation of the large
intestine, requires pretreatment, is invasive, and places a heavy burden on patients with
respect to their time and the physical, and financial perspectives, and thus, is difficult to be performed colonoscopy frequently.1-3)
Therefore, non-invasive and inexpensive biomarkers that can easily assess the inflammatory
bowel disease (IBD) activity are required. Therefore, a non-invasive, simple, and
relatively inexpensive fecal calprotectin test is used as a diagnostic aid.4)
Calprotectin is a highly stable protein that is produced by neutrophils, a type of white
blood cell, and reflects the inflammatory state of the intestinal mucosa.5) The
results of the fecal calprotectin test show a positive correlation with the results of
colonoscopy.1) Thus, the need to take laxatives, which is required for
colonoscopy, is eliminated. Patients just need to collect a small amount of stool in a
dedicated specimen collection container at home or elsewhere and send it to their
physicians or a designated testing institution.
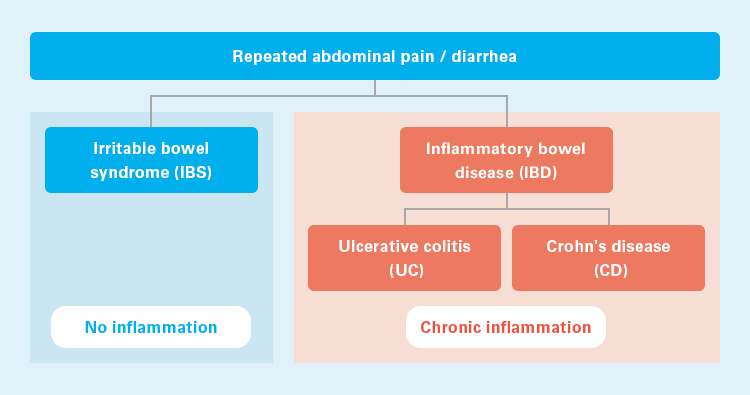
Abdominal pain and diarrhea, which are the major symptoms of IBD, can also be seen in
irritable bowel syndrome (IBS), which makes it difficult to differentiate between the two
diseases in their early stages. Since the treatment for IBD and IBS is different, it is
necessary to differentiate between these two diseases. While IBD is associated with
inflammation of the intestine, no inflammation is observed in IBS; therefore, IBD and IBS
can be differentiated on the basis of the presence of inflammation.6)
The National Institute for Health and Care Excellence (NICE) Diagnostic guidance [DG11]
recommends fecal calprotectin test as an aid to differential diagnosis between IBD and
IBS.7) Fecal calprotectin test is an effective biomarker for differentiating
IBD from IBS,8) and The NICE Clinical guideline [CG61] clearly states that
invasive colonoscopy is not necessary for the definitive diagnosis of patients who meet
the diagnostic criteria for IBS.9)
Similarly, the European Crohn’s and Colitis Organisation (ECCO) Guideline states that the
results of the fecal calprotectin test are in good agreement with those of colonoscopy in
terms of assessment of disease activity, and that it is an important test in various
clinical settings, including initial diagnosis and monitoring of disease actitivity.10)
IBD and IBS11)
In the recent years, the goal of IBD treatment has been to reduce the disease activity and
achieve mucosal healing based on endoscopic remission,4) and therefore, it has
become important to actively monitor the disease activity.
The fecal calprotectin test has been reported to be very useful as a diagnostic aid, which
can easily assess the disease activity in IBD, such as relapse and remission, by utilizing
the correlation between fecal calprotectin concentration and the degree of inflammation.4,
12)
The ECCO Guideline recommends the implementation of the fecal calprotectin test, which is
assessed as follows:10)
(1) Validity [correlation with gold standard]: Good,
(2) Responsiveness to changes in condition: Good,
(3) Signal-to-noise ratio [ability to differentiate changes in condition from background
variability]: Moderate, and
(4) Practicality: High.
The fecal calprotectin test is not a substitute for colonoscopy, which is a definitive test for IBD, particularly for understanding the disease activity of ulcerative colitis (UC). However, the fecal calprotectin can be performed frequently as it is non-invasive and may predict relapse earlier.13-15) Additionally, it may reduce the number of colonoscopies performed.
References
Inflammatory bowel disease (IBD) is any disease that causes inflammation in the digestive
organs. IBD in a broad sense includes infectious enteritis and drug-induced enteritis.
Typically, however, ulcerative colitis (UC) and Crohn's disease (CD) are collectively
referred to as IBD.
The number of patients with IBD worldwide continues to increase. The incidence of IBD was
estimated to be about 6.8 million patients in 2017, and the prevalence per 100,000
population had also increased from 79.5 patients in 1990 to 84.3 patients in 2017.1)
IBD was a commonly observed disease in the United States and Western Europe. However, the
number of patients has recently been increasing in Asian countries such as India and
Japan,2) and IBD has become one of the most important diseases worldwide.
Although it is not a life-threatening disease, appropriate treatment from an early stage
is necessary, since it adversely affects the quality of life (QOL)3) and
activities such as studying4) and working5); further, IBD has been
reported to be associated with colorectal cancer.6)
At present, IBD cannot be completely cured by medication. The goal of treatment is to
reduce the disease activity, actively assess the disease activity with colonoscopy, and
achieve mucosal healing based on endoscopic remission.7-8)
Annual trends in the number of patients with IBD and prevalence per 100,000 population1)
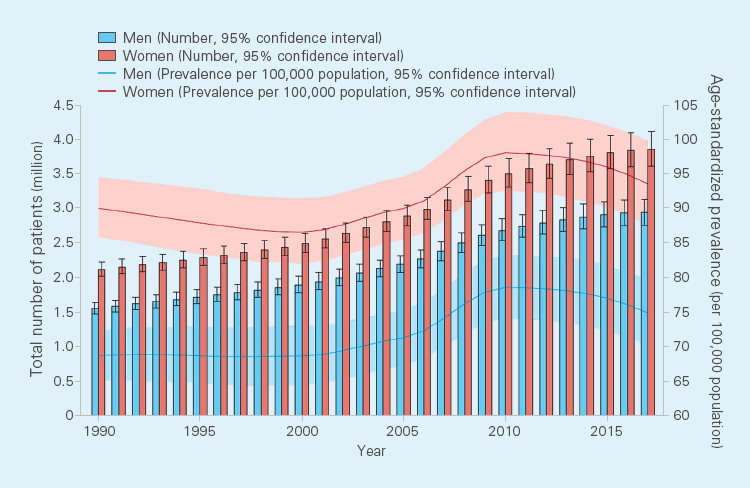
From GBD 2017 Inflammatory Bowel Disease Collaborators.: Lancet Gastroenterol Hepatol. 2020; 5(1): 17-30. Which is Open Access article under the CC BY 4.0 license.
References